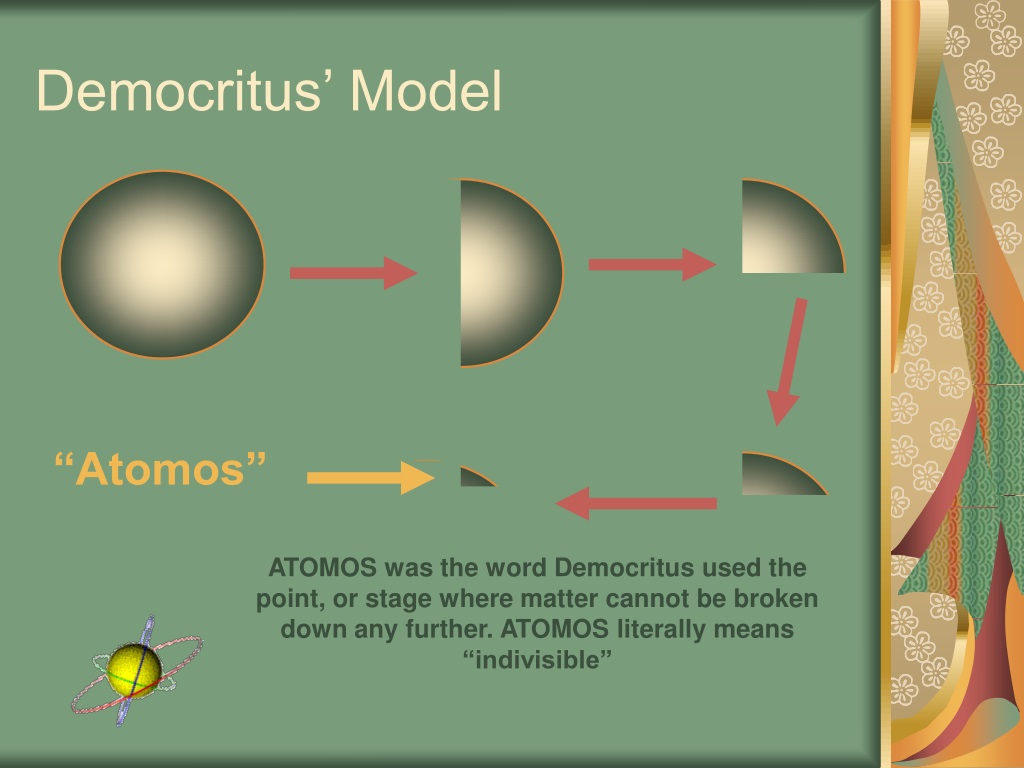
From the concept of orbitals, one explains molecular structure, chemical bonding, hybridization etc and also things like spectrum of various materials. Previously, we defined an atom as the smallest part of an element that maintains the identity of that element. 1 ), is a fundamental concept that states that all elements are composed of atoms. In the Thomson atom model electrons are embedded as little particles in a positively charged mass like raisins are embedded in a cake. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 4.2.1 4.2. Only when the idea of electrons as particles in atoms was introduced, attempts were made to understand chemical bonding and other physical and chemical properties.Īs of now, the electron cloud model which incorporates the principles of quantum mechanics is the most successful. The discovery of the electron led Thomson to the development of a first atomic model that would include a subatomic particle. These models were used to explain and hence to understand properties of matter. It was soon realized that the model was flawed and soon Bohr put up his model in which he used Quantum concepts of Planck and Einstein to explain stability of matter and the Hydrogen spectrum.Īnd they process continued when Sommerfeld proposed his model. The theory of Democritus held that everything is composed of 'atoms,' which are physically, but not geometrically, indivisible that between atoms, there lies empty space that atoms are indestructible, and have always been and always will be in motion that there is an infinite number of atoms and of kinds of atoms, which differ in shape and size. 
In his model, he took the electrons embedded into the positive sphere because he found that these electrons (not some positively charged particle) were emitted by a vast range of marerials.įurther, when Lord Rutherford discovered that the positive charge and most of the mass was concentrated in a small region called the nucleus, the proposed his model based on what evidence was available at that time on properties of atoms. You learned earlier how all matter in the universe is made out of tiny building blocks called atoms. 
4.2: Indivisible - The Atomic Theory is shared under a CK-12 license and was authored, remixed, and/or curated by Marisa Alviar-Agnew & Henry Agnew. However, they should be able to explain the basic properties of matter.įor example, when the electron was first discovered, then JJ Thompson proposed the plum cake model in which he took into consideration the two main points -ġ) From the CRT experiments, it was clear that all matter had those electrons within then thus it would be logical to suppose that electrons were a part of atoms.Ģ) In order to explain the charge neutrality, he supposed that the sphere in which electrons are embedded is positively charged. In chemical reactions, atoms are combined, separated, or rearranged. 1: Lavoisiers discovery of The Law of Conservation of Matter led to the Laws of Definite and Multiple Proportions and eventually Daltons Atomic Theory. This is because his theory provided a logical explanation of concepts and led the way to new experimentations. A Video Introduction to Atomic Theory through the Nineteenth Century From Crash Course Chemistry. Such models would be purely theoretical constructs. Daltons atomic theory or model was accepted despite errors. When the exact structure of the atom remained quite unknown, models were proposed based on experimental evidence of the properties of matter. \), control of individual atoms can be use used create animations.An atomic model is a theory trying to explain the structure of the atom.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
